41 health claims on food labels are standardized and regulated
Unit 6 Nutrition Labeling(Nutrition labeling; dietary ... - Quizlet percentages of daily value are based on the recommended daily allowances-amount of nutrients a person should get each day.These numbers refer to the DV(daily value) that one serving of the food provides as a percentage of established standards. ex.a label can show that a serving of the food provides 30% of the daily recommended amount of fiber. eCFR :: 21 CFR Part 101 -- Food Labeling § 101.1 Principal display panel of package form food. The term principal display panel as it applies to food in package form and as used in this part, means the part of a label that is most likely to be displayed, presented, shown, or examined under customary conditions of display for retail sale. The principal display panel shall be large enough to accommodate all the mandatory label ...
Labeling Policies | Food Safety and Inspection Service Jun 17, 2022 · Final rule amending the nutrition labeling regulations to change the definition of "meal-type" products to allow for nutrient content claims on multiple-serve food containers, to adopt the definition of "main dish" used by the Food and Drug Administration (FDA), and to define how meal-type products and main dishes should be nutrition labeled.

Health claims on food labels are standardized and regulated
Packaging, Labeling, Transporting, Storing — Food Law Qualified Health Claims provide health claims based on less scientific evidence than is required for a Health Claim, as long as the Qualified Health Claims does not mislead the consumers." Function and Structure Claims: "Dietary supplement labels may bear statements that describe the role of a nutrient or dietary ingredient intended to affect ... Label Claims for Food & Dietary Supplements | FDA Among the claims that can be used on food and dietary supplement labels are three categories of claims that are defined by statute and/or FDA regulations: health claims, nutrient content claims,... eCFR :: 21 CFR Part 130 -- Food Standards: General The Code of Federal Regulations (CFR) is the official legal print publication containing the codification of the general and permanent rules published in the Federal Register by the departments and agencies of the Federal Government. The Electronic Code of Federal Regulations (eCFR) is a continuously updated online version of the CFR. It is not an official legal edition of the CFR.
Health claims on food labels are standardized and regulated. Bayblend® T85 XF - Covestro food processing, food contact and others). If the intended use of the product is for the manufacture of a pharmaceutical, medical device or of intermediate products for medical devices or for other specifically regulated applications which may lead to a regulatory obligation of Covestro, Covestro must be contacted in advance to provide its agreement to sell such product for such purpose. CH 2 - NUTRITION Flashcards - Quizlet Structure/function claims on dietary supplement labels are not regulated as rigorously as health claims. TRUE. Serving sizes are standardized on food labels to allow consumers to compare products. ... Nutrient content and health claims are approved by the: FDA. A _____ claim refers to a relationship between a nutrient, food, or dietary ... chapter 2 true and false Flashcards - Quizlet food companies are prohibited from using nutrient or health claims in food labels that are not approved by the FDA false Yogurt parfaits offered at some fast food restaurants are a more healthful choice than fruit true the number of servings of each food group recommended in the USDA food patterns is based on age, gender, and activity level false Code of Federal Regulations Title 21 - Food and Drug Administration The name of a substitute food that complies with all parts of this regulation is the appropriate expressed nutrient content claim and the applicable standardized term. (f) Label declaration. (1) Each of the ingredients used in the food shall be declared on the label as required by the applicable sections of part 101 of this chapter and part 130.
FDA Regulation of Product Claims in Food Labeling - JSTOR food labeling, not just to claims relating to composition or nutritional value. There is no established taxonomy for food labeling claims, but they can be conveniently broken down into the following 14 categories: 1. Extrinsic claims (e.g., price, packaging, and convenience). 2. Utility claims (e.g., recipes and methods of using the food PDF Making Smart Choices: Health Claims, Regulation, and Food Packaging HEALTH CLAIMS, REGULATION, AND FOOD PACKAGING . by . Kimberly Mei Ling Butler. BS, James Madison University, 2002 ... and led the Food and Drug Administration to consider mandating a standardized, national ... 52% of consumers refer to food labels before purchasing; 85% for a first time purchase (Geiger, 1998; Morreale & Schwartz, 1995). ... Using Dietary Supplements Wisely | NCCIH However, some types of claims related to health or the way that the product affects the structure or function of the body may appear on dietary supplement labels. Health claims describe a relationship between a substance in the supplement and reduced risk of a disease or condition. They must be based on scientific evidence. The Food Labeling Modernization Act Introduced in Congress (Again) For added sugars, FDA would need to promulgate a regulation that would set an added sugars level that would disqualify the food from making any health claims. The bills would allow FDA to request documentation from a manufacturer on the substantiation for its health claims, after which the manufacturer would have 90 days to comply.
Large-scale GWAS of food liking reveals genetic determinants and ... 18.5.2022 · We present the results of a GWAS of food liking conducted on 161,625 participants from the UK-Biobank. Liking was assessed over 139 specific foods using a 9-point scale. Genetic correlations ... Trans fat - Wikipedia Trans fat, also called trans-unsaturated fatty acids, or trans fatty acids, is a type of unsaturated fat that naturally occurs in small amounts in meat and milk fat. It became widely produced as an unintentional byproduct in the industrial processing of vegetable and fish oils in the early 20th century for use in margarine and later also in snack food, packaged baked goods, and for frying fast ... Nutrition chap 12 quiz Flashcards | Quizlet A. A nutrient claim is. A. a declaration on food packages to indicate a possibly beneficial nutrient. B. not federally regulated. C. more specific information about what the food product contains. D. a statement on the label about how that nutrient may affect the human body's structure. E. All of the above. Aromatherapy | FDA Certain citrus oils used safely in food can also be harmful in cosmetics, particularly when applied to skin exposed to the sun. FDA doesn’t have regulations defining “natural” or “organic ...
Federal Register :: Clinical Trials Registration and Results ... 21.9.2016 · It implements the provisions of section 402(j) of the Public Health Service Act (PHS Act) (42 United States Code (U.S.C.) 282(j)) as amended by Title VIII of FDAAA and including technical corrections made to FDAAA under Public Law 110-316), which were intended to improve public access to information about certain clinical trials of U.S. FDA-regulated drugs, biological products, and devices ...
Introduction - Front-of-Package Nutrition Rating Systems and Symbols ... Food packages have long included nutrition messages to consumers, whether to provide the consumer with nutrition information or to help market food products. The Nutrition Labeling and Education Act of 1990 amended the Federal Food, Drug, and Cosmetic Act to require that labels of most packaged food products regulated by the Food and Drug Administration (FDA) include a standardized nutrition ...
FDA Food Labeling Enforcement - LabelCalc Intentionally deceiving the public by including inaccurate health or nutrition claims, for instance, could result in a significant fine. While I don't personally know anyone who has gotten to the point of being fined for non-compliance, it does happen, so it's a good idea to make sure you comply with nutrition labeling guidelines.
19 which claims are not closely regulated by the food - Course Hero The US Food and Drug Administration (FDA) has the power to regulate health claims on food labels under the Nutrition Labeling as well as Education Act of 1990. These assertions address the relationship between key nutrients or chemicals found in food as well as a specific disease or health problem. FDA regularly monitors nutrient content claims.
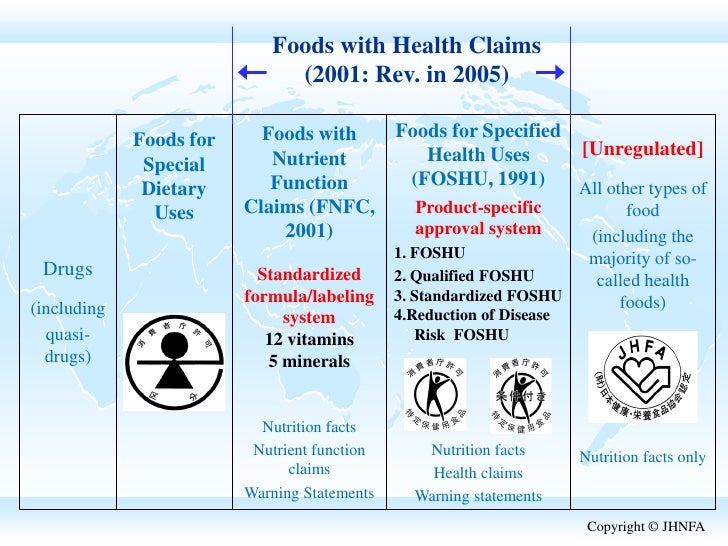

Post a Comment for "41 health claims on food labels are standardized and regulated"